 Our research group, in collaboration with a Hungarian research team, just published a new study on the effects of uridine and exogenous ketone supplements in a rat model of epilepsy and showed for the first time that uridine enhance the anti-epileptic effects of ketosis.
Our research group, in collaboration with a Hungarian research team, just published a new study on the effects of uridine and exogenous ketone supplements in a rat model of epilepsy and showed for the first time that uridine enhance the anti-epileptic effects of ketosis.
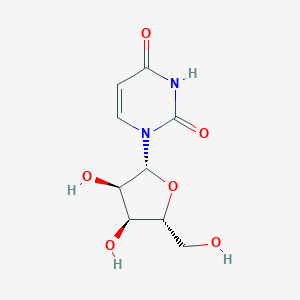
Uridine is an intriguing molecule that the body produces naturally in the liver and is delivered to other tissues via circulation. If you think back long and hard to high school biology, you might recall uridine as one of the five nucleosides that compose our genetic material. Uridine is taken up by the brain and is used for RNA synthesis and provides building blocks for healthy cell membrane constituents. Consequently, it plays an important role in brain health and cognition. There is research to support the potential of uridine supplements, sold as “uridine monophosphate” to increase neuronal plasticity and synapse formation, which are required for proper learning, memory, and cognition. Importantly, and regarding the topic at hand, uridine has been shown to have anticonvulsant effects in different models of epilepsy
Of course, if you are familiar with the history of the ketogenic diet, you will know that it was used in the early 1900s as the standard of care treatment for epilepsy, and it is used today as an effective treatment in children, adolescents, and adults with epilepsy and other seizure disorders. More recent research suggests that exogenous ketone supplements may offer an alternative metabolic therapy to the ketogenic diet to treat different brain disorders, including psychiatric disorders, too, partly due to the effects of ketone bodies on adenosine and adenosine receptors.
Adenosine receptor activity plays a significant role in neuronal excitability and seizure activity and may be partly responsible for the anticonvulsant effect of ketosis. Previous findings suggest uridine may also interact with adenosine receptors and reduce neuronal excitability. In addition, both ketone bodies and uridine may increase the level of GABA in the brain, which has anticonvulsant effects. Taken together, we speculated that it is possible that the co-administration of both may enhance their beneficial effects on seizures. The advantage of administering two complementary compounds that increase the efficacy of the other is that it reduces the side effects of having to use high doses of these compounds individually. Furthermore, optimizing the effectiveness of these supplements would allow more flexibility to patients following a ketogenic diet, as it would allow patients to follow a more liberal ketogenic diet, thereby increasing compliance and tolerability.
To test this hypothesis, we studied the effects of co-administering uridine and exogenous ketone supplements in a rat model of human absence epilepsy. This model has a genetic predisposition to absence epilepsy and experiences spontaneous spike-wave seizures (a hallmark of absence epilepsy) on electroencephalographic (EEG) recordings.
The exogenous ketone supplement we used was a combination of beta-hydroxybutyrate (BHB) salts with medium chain triglyceride (MCT) oil. To measure the effect, EEG recordings were taken, and based on these recordings, we could see if the rats experienced a spike-wave seizure.
What did we find?
This study revealed two important findings:
1. The anticonvulsant effect of uridine is due, at least partly, through the A1 adenosine receptor
2. A low dose of uridine strengthened the anticonvulsant effect of exogenous ketones
Our results confirmed that uridine alone decreased seizure activity. Importantly, this study revealed that the adenosine receptor, A1R, is necessary for the anticonvulsant effect of uridine, but not A2AR. We confirmed this by administering their respective adenosine receptor antagonists (blocks adenosine receptor activity) alongside uridine and found that the effects were abolished only when A1R receptors were inhibited. The A1 adenosine receptor has traditionally been thought to have neuroprotective effects whereas the A2A adenosine receptor has been implicated in aging and neurotoxicity. However, this is the first study to evaluate their influence on uridine and exogenous ketones in a model of epilepsy.
Uridine Treatment:
Uridine + Adenosine receptor antagonist:
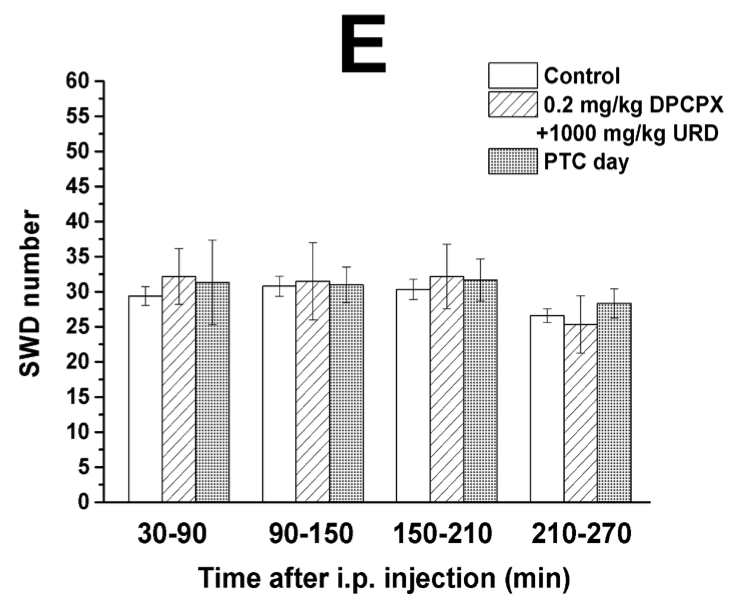
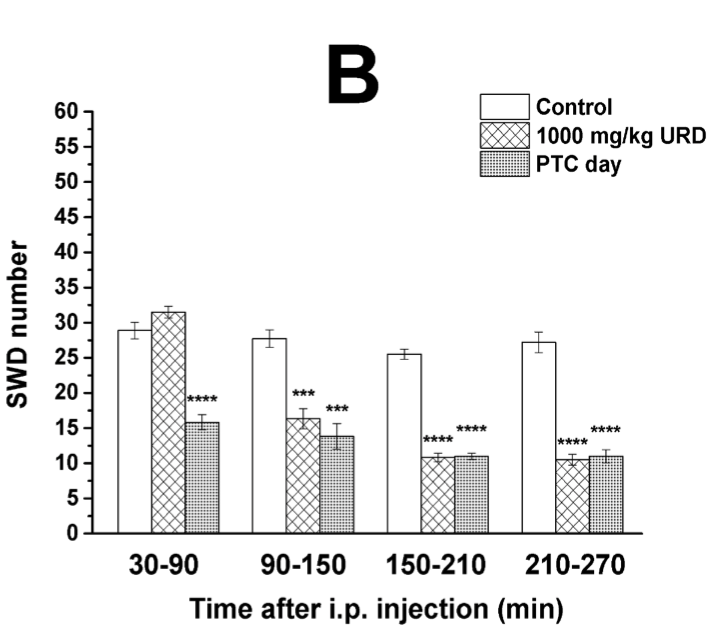
Our next novel finding was that a lower dose of uridine did not significantly reduce seizure activity unless paired with the ketone supplement. Or, put another way, uridine enhanced the alleviating effects of ketosis.
Low dose uridine + Exogenous ketones:
It is possible that the synergistic effect may be due to uridine’s interaction with adenosine and uridine receptors, in combination with BHB-induced increase in adenosine levels and adenosine receptor activation. Consequently, these effects decrease neuronal excitability and activity, thereby reducing absence epileptic activity.
Since uridine is a molecule the body produces naturally, proper dosing uridine supplementation could represent a safe treatment of epilepsy with minimal to no side effects and reduced risk compared to some pharmacological therapies. Its safety has been previously reported, and findings have shown that it is a well-tolerated drug with only minor toxic potential. Exogenous ketone supplements are also well-tolerated and safe and may have therapeutic potential in the treatment of epilepsy. Since the co-administration of uridine and exogenous ketones enhanced the beneficial effect of each other, this combination represents a novel treatment for absence epilepsy, but further animal and clinical studies are needed. Lastly, this study only assessed these treatments after a single dose, but the results suggest that long-term co-administration may be even more effective, which should be the focus of future research.
Written by: Kristi Storoschuk; Edited by: Dominic DAgostino, Csilla Ari Dagostino